Projects in the lab use a multidisplinary approach combining synthetic carbohydrate chemistry, enzymology, membrane protein biophysics and molecular biology to study the structure, biosynthesis, membrane transport, and functions of exopolysaccharide that make up the extracellular matrix of bacterial biofilm communities.
Research Area #1
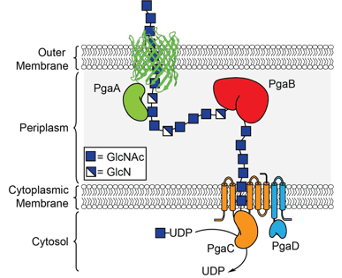
We are interested in developing tools to study the biosynthesis of bacterial exopolysaccharides and the role of these exopolysaccharides in biofilm formation and bacterial infections.
Research Area #2
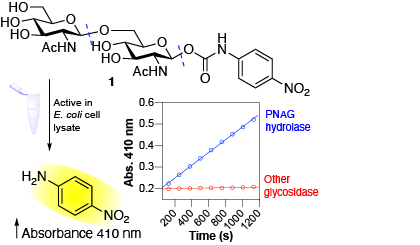
We are interested in identifying and characterizing enzymes involved in the breakdown of bacterial biofilm exopolysaccharides and studying the role of these enzymes in biofilm dispersal.
Research Area #3
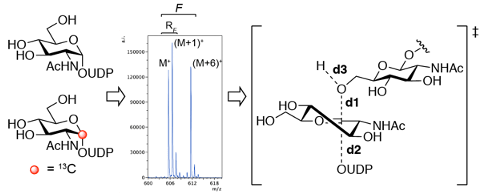
We are developing chemical probes that disrupt exopolysaccharide biosynthesis in order to study their role in biofilm assembly in vivo. This approach combines mechanism based inhibitor design and transition state analog design approaches based on KIE measurements to develop inhibitors of the glycosyltransferase enzymes involved in exopolysaccharide biosynthesis.